Intravenous Vitamin C and Cancer a Systematic Review
ane. Introduction
Vitamin C is an essential micronutrient, that plays an important role in numerous physiological processes in the human being torso. Different nearly mammals, humans lack the ability to generate endogenous vitamin C due to a mutation in the GULO cistron and are thereby completely dependent on dietary intake. The biological efficacy of vitamin C depends on its redox abilities and it functions as a cofactor in many enzymatic reactions. In physiological concentrations, it besides functions every bit an antioxidant.
Past the 1970s, Nobel Price winner Linus Pauling had already developed a strategy to use intravenous (IV) vitamin C in cancer patients [one,two]. He treated patients with advanced cancer with high doses of vitamin C and reported a positive effect on survival. Nevertheless, these studies have been methodologically criticized on several aspects such as data collection and information analysis. This resulted in a limited use of vitamin C in cancer patients. Other studies performed afterwards could also not reproduce these results; however, opposed to the intravenous use of vitamin C by Pauling et al, in nigh of these studies, oral vitamin C supplementation was used [3]. Pharmacokinetic studies show that the fashion of administration makes a big difference equally peak plasma vitamin C concentrations after intravenous administration are much higher (up to 70-fold) than after oral intake [4]. Summit plasma concentrations as well keep to increase when the intravenous dose of vitamin C is increased, while peak plasma concentrations plateau effectually 220 µM even though oral doses are increased.
In that location are multiple hypotheses almost the mode vitamin C has anti-tumor effects. An important possible machinery of action is that in pharmacological concentrations (especially after intravenous use) vitamin C functions as a pro-oxidant and stimulates the formation of hydrogen peroxide. This hydrogen peroxide can create reactive oxygen species (ROS), that directly have cytotoxic activity on cancer cells [v]. Another important hypothesis is that vitamin C can create important epigenetic changes due to its effect on 2-oxoglutarate-dependent dioxygenases, like histone and DNA demethylases [6]. In preclinical studies investigators as well show that vitamin C tin have a synergetic issue with some types of chemo- and immunotherapy [7,8,nine,10,11].
Additionally we showed in pre-clinical studies that vitamin C has an important role in the immune system, every bit information technology stimulates the production and/or activation of immune cells, like T-lymphocytes and natural killer cells, that accept a function in fighting against pathogens and cancer cells [12,13,fourteen].
In our previous inquiry on vitamin C we noticed that many of our patients receiving intensive chemotherapy and/or stem jail cell transplantations for hematological malignancies have low vitamin C plasma concentrations [15]. This could be the result of low dietary intake of these patients or of an increased demand for vitamin C in tumor cells or in immune cells. In extension of our results, other researchers observed that low vitamin C plasma levels in patients with various types of avant-garde cancer were associated with worse survival [16].
Patients that receive intensive chemotherapy and/or stem prison cell transplantations are prone for infectious complications. Boosting their allowed organisation with vitamin C to hasten immune recovery and thereby foreclose infectious complications is attractive, since vitamin C is cheap and generally available. However, since some vitamins have been shown to promote cancer evolution, we were interested in the effects of vitamin C on cancer progression and its safety. To this end, we conducted a systematic review of the literature on vitamin C administration in cancer patients. We focused on administration road, efficacy and on the side-furnishings in combination with or without other cancer treatment.
2. Materials and Methods
two.one. Objectives
The aim of this review is to assess the effectiveness of vitamin C in the handling of cancer, with or without adjuvant standard anti-cancer handling like chemotherapy and radiotherapy.
We researched the literature on the following hypotheses:
-
— Vitamin C administration is more effective in the treatment of cancer than placebo or no handling in susceptible populations.
-
— Different routes of vitamin C administration (intravenous/oral) may differ in effectiveness in treating cancer.
The reached serum and/or tissue vitamin C concentrations with supplementation were also of involvement and were noted when given.
An try was also made to quantify toxicity and side effects of vitamin C and the findings were considered in the discussion to determine the risk–benefit ratio of the treatment.
ii.2. Protocol and Registration
This systematic review was written conform the PRISMA statement for reporting systematic reviews of studies that evaluate wellness intendance interventions [17]. It is registered with the University of York Eye for Reviews and Dissemination International Prospective Register of Systematic Reviews.
2.iii. Eligibility Criteria
2.three.1. Types of Studies
Studies on the consequence of vitamin C administration in cancer patients after diagnosis were included. All study designs were allowed except for Phase I trials and instance reports, since there was a lack of all-encompassing randomized controlled trials (RCTs) merely the quality of the studies was weighted during analysis and word. Linguistic communication was restricted to English.
two.3.2. Types of Participant
Inclusion criteria
— Studies with patients of all ages and both genders with all types of diagnosed cancer.
Exclusion criteria
— Studies investigating the effect of nutritional supplements.
— Studies on the effect of vitamin C administration in the prevention of cancer.
two.3.3. Types of Intervention
Studies on the upshot of clinical vitamin C administration, every bit mono-therapy or in combination with other standard cancer treatment regimes. The dose and way of delivery were considered in subgroup analyses.
2.3.iv. Types of Outcome Measures
Primary outcome measure out was overall survival. Secondary outcome measures were progression-free survival, tumor response, response rate, disease-free survival, adverse effects, quality of life (QOL), clinical response and performance status (PS).
two.4. Literature Search
Studies were identified past searching the PubMed and EMBASE databases and snowballing from review articles and relevant studies. The terminal search was run on the 11th of March 2019.
The following search terms were used to deport the search: Neoplasms; Cancer; Malignancy; Leukemia; Lymphoma; Ascorbic acid; Vitamin C; Ascorbate; Dehydroascorbic acid; Randomized controlled trial; RCT; Randomized; Controlled clinical trial; Prospective study; Clinical trial; Instance-command; Accomplice; Phase 2; Observational study; Reduced infection; Overall survival; Progression-gratis survival; Toxicity; Quality of life; Tumor response; Response charge per unit; Affliction-gratis survival. The complete search strategy is shown in Supplementary File S1. Bibliographies of identified articles were besides reviewed and searched manually for additional references.
2.5. Data Collection and Assay
Cess of eligibility of the articles for inclusion in this review was performed and peer reviewed by two of the authors. The identified articles were screened on title and abstract in agreement with the inclusion and exclusion criteria by Due east. Lookermans (E.L.), who discarded studies that were clearly ineligible but aimed to be overtly inclusive rather than risk losing relevant studies. Subsequent total text assessment resulted in the last written report choice.
Data were collected by E. L. and peer reviewed past Grand. van Gorkom (M.G.) with employ of a information extraction canvass based on the Cochrane Consumers and Communication Review Group's data extraction template [18]. For each included report, information was extracted regarding the methods of the study (aim, study pattern, number of groups), the participants (number of patients, patient clarification, geographic location, methods of recruitment, inclusion criteria for participation, exclusion criteria for participation, age, gender) the intervention (vitamin C treatment, dose, schedule, mode of delivery, boosted treatment, previous cancer treatments received, setting) and the outcomes and comparison groups (main effect measures, secondary event measures, method of assessing outcome measures, method of follow-up for non-respondents, event assessment, length of follow-up, frequency, relevant agin events). Discrepancies in the data extraction were resolved through discussion.
The quality of all eligible studies was assessed independently by two authors (Eastward.L. and M.G.) with apply of predefined risk of bias criteria, with discrepancies resolved by discussion and a third writer (G.B.) when necessary. For randomized-controlled trials "The Cochrane Collaboration's tool for assessing risk of bias in randomized trials" was used [xix], for non-randomized comparing studies (studies were there is a grouping of patients included without vitamin C) the ROBINS-1 tool was used, a Cochrane risk of bias assessment tool for non-randomized studies of interventions [20] and the Effective Public Wellness Do Project (EPHPP) Quality Assessment Musical instrument was used for not-comparing studies [21]. These tools were used to make judgments about the extent of bias that may be present in each of the studies and to rate the data in each component of the paper (Supplementary File S2).
three. Results
3.1. Written report Option
A total of 975 articles was retrieved by the PubMed and EMBASE databases search. An additional article was found through article references, bringing the total number of records suitable for further evaluation to 976. Later removal of duplicates there were 920 articles left for investigation. By scanning the title and abstruse of these records, 882 records were excluded because they clearly did not lucifer the inclusion criteria (mostly it were preclinical studies). Thirty-eight manufactures were evaluated on their total text. Of these articles, 19 records were excluded based on the inclusion criteria. In most of these articles, vitamin C supplementation was non the primary intervention, but combined with other experimental treatments, like other vitamins or arsenic trioxide. This resulted in 19 records being included for qualitative synthesis (Figure i).
iii.2. Study Characteristics
The study characteristics of the 19 manufactures selected for this review are described in Table 1.
3.two.ane. Participants
The number of participants in these studies ranges from 14 to 1826.
All studies described different individual patients except the 2 studies past the same authors. In both of these studies, 100 patients with terminal cancer which were treated with IVC were compared to one thousand similar control patients. The second report 10 of the original studied patients were replaced since at that place were not enough suitable control patients, merely ninety patients and most of the controls were identical to the first written report [1,23].
Nearly trials included patients with a multifariousness of cancer types, and virtually of the time patients were in a final or advanced stage. In only 6 studies a specific cancer type was treated: ii times chest cancer, 1 ovarian cancer, 1 colorectal cancer, 1 prostate cancer and 1 acute myeloid leukemia.
All trials included both sexes, except for the studies on chest cancer and ovarian cancer that logically treated only women and the study on prostate cancer that logically treated only men. The average age of the participants was approximately 60 years.
iii.2.2. Intervention
In 8 studies, vitamin C was given intravenously (IV), all in different doses and time intervals [22,25,26,27,28,33,35,37]. In 8 studies intravenous vitamin C (IVC) was given followed by or in combination with oral vitamin C supplementation [one,2,23,24,30,31,34,36]. In iii studies vitamin C supplementation was only prescribed orally [3,29,32].
3.ii.3. Other Treatments
In 7 studies, conventional anti-cancer treatment was given in addition to the administration of vitamin C [22,24,25,27,34,35,37]. In 6 studies, this was not specifically documented, but it seems unlikely that patients had concomitant treatments [iii,26,28,30,32,36] and in half dozen articles it was written that no boosted treatment was given at the time of the intervention with vitamin C [1,ii,23,29,31,33]. In iv articles, the researchers described that patients received conventional cancer therapy prior to participation [2,31,32,35], 13 articles study no information of previous treatments [1,3,22,23,24,25,26,27,28,thirty,33,34,36] and in ii studies patients received no prior treatment [29,37].
3.2.4. Outcome Measures
Ten articles discuss the effect of vitamin C on overall survival [one,three,23,24,26,27,29,30,32,37], 9 manufactures the consequence on clinical response in general [2,25,26,28,29,30,31,33,37], 7 on QOL and PS [3,26,30,31,34,35,36] and 14 articles report on the safety and toxicity of vitamin C treatment [2,3,22,25,26,27,29,30,31,33,34,35,36,37].
three.3. Risk of Bias in Included Studies
Figure 2 presents the bias risk assessment as percentages across all RCTs. Bias judgment was based on "The Cochrane Collaboration'southward tool for assessing risk of bias in randomized trials". Supplementary File S3 shows a summary of the risk of bias assessment of each item for each included RCT.
In all RCTs, the selection process and the randomization procedure were not clearly described. It was therefore impossible to make a statement nearly the selection bias in the studies. Two of the 4 studies were blinded. There was a low risk of detection bias since the primary outcome in the RCTs was overall survival. There did not seem to be other forms of bias in the selected RCTs.
The ROBINS-I tool, used for the non-randomized comparative studies, showed moderate to loftier adventure of bias for the majority of the comparative studies as seen in Table 2. The Effective Public Health Practice Projection tool, used for the not-randomized not-comparative studies, showed moderate to weak quality of the majority of non-comparative studies equally seen in Tabular array three. Although certain studies used wide option criteria, choice bias is hard to avoid without randomization.
iii.four. Results of Private Studies
Results of the individual studies are summarized in Tabular array 1.
3.4.1. Overall Survival
Ten of the included studies measured overall survival in vitamin C treated patients [1,3,23,24,26,27,29,30,32,37] (Table 1). All studies compared their results with those obtained with a control group. In three studies, no consequence of vitamin C on survival time was observed [three,29,32]. Two of those studies were RCTs. In seven studies, the researchers found a positive effect of vitamin C on survival time [1,23,24,26,27,30,37], 2 of those studies were RCTs. 1 of these RCTs was washed in acute myeloid leukemia in a small-scale group of patients treated with a hypomethylating agent (decitabine) that in vitro has a synergistic effect on vitamin C on TET2 expression, apoptosis and proliferation of tumor cells. Patients received a relatively depression dose of vitamin C intravenous and median overall survival increased with half-dozen months [37]. In the other RCT, the overall survival also trended toward improvement with vitamin C addition to standard chemotherapy for ovarian cancer, only since the patient groups were very pocket-sized the increase in median overall survival was non meaning [27].
Intravenous versus Oral Vitamin C
All studies in which researchers suggested a positive effect of vitamin C on survival fourth dimension, supplementation was administered intravenously, with [1,23,24,xxx] or without [26,27,37] oral supplementation. In the 3 studies, in which no upshot of vitamin C on survival was observed, the supplementation was administered only orally [3,29,32].
three.4.two. Clinical Response
The consequence of vitamin C on clinical response was assessed in 9 studies [ii,25,26,28,29,30,31,33,37], of which 6 showed at least some clinical improvement [two,25,26,28,30,37]. In the kickoff written report in patients with advanced stage various types of cancer, in which some positive effects were described, 10% of the patients experienced tumor regression [2]. This regression, yet, was mostly measured based on clinical findings (and not imaging) and earlier tumor progression or metastasis was not e'er histologically proven. Other clinical improvements observed in that report were as well highly subjective.
When IVC was given in 14 patients with diverse types of advanced cancer in combination with cytotoxic chemotherapy, 43% of the patients experienced a transient, simply sometimes long-lasting stable illness; the investigators thought this was the effect of the vitamin C since it was highly unlikely to exist due to chemotherapy alone [25]. All the same, no tumor regression was seen. In 15 patients with os metastasis IVC had a positive effect on relief of pain [26]: with IVC 53% of patients had an significant comeback of the pain versus xiii% with chemotherapy and 0% in controls without treatment. Another research group also observed a decrease in pain and decreased use of narcotic drugs [30]. They also saw some subjective other clinical improvements possibly related to the handling with vitamin C.
In 75% of xx prostatic cancer patients, PSA (prostate-specific antigen) decreased after the assistants of IVC [28]. This issue, all the same, was non confirmed in another trial were none out of 23 patients with prostate cancer experienced a reduction of the PSA [31].
In 2 other studies there was too no objective clinical improvement [29,33]. In 1 of these (an RCT) 64% of vitamin C patients claimed relief of pre-treatment symptoms, just this was like in the placebo grouping (65%) [29].
The merely objective improvement of clinical response was seen in patients with astute leukemia; patients with vitamin C had significantly ameliorate complete response rates subsequently chemotherapy than without [37].
Intravenous Versus Oral Vitamin C
All investigators that reported positive effects of vitamin C on clinical response administered vitamin C 4, with [2] or without [25,26,28,37] oral vitamin C.
3.4.three. Quality of Life and Performance Status
QOL and/or PS afterward vitamin C handling were assessed in vii of the included studies [3,26,30,31,34,35,36]. In 5 studies, investigators showed a beneficial issue of vitamin C [26,30,34,35,36] while in 2 studies no benefit of vitamin C could be demonstrated [3,31].
Remarkable is ane RCT in which 63% of the 60 patients with various types of advanced phase cancer on vitamin C claimed some comeback in symptoms but also 58% of the patients on placebo [3].
Intravenous Versus Oral Vitamin C
Vitamin C was given IV (+/- orally) in the studies in which the investigators demonstrated a beneficial outcome of vitamin C on QOL and PS [26,30,34,35,36]. In 1 written report in which no effect was seen vitamin C was given intravenously in combination with a low dose of vitamin C orally, in the other study without benefit only oral supplementation was used [three,31].
3.4.iv. Safety and Toxicity
Fourteen articles study on the safe and toxicity of vitamin C handling [two,3,22,25,26,27,29,thirty,31,33,34,35,36,37]. In all studies, no vitamin C related toxicity was observed. In 10 studies, possible side effects of vitamin C were seen just these were generally mild, and in general non more than in a control grouping. In ane of these studies in which vitamin C was given through continuous infusions, the investigators described 2 serious agin events (SAEs) that were peradventure related to vitamin C treatment: kidney stones and hypokalemia [33]. In ii studies, no vitamin C related side furnishings were observed [27,35]. Investigators of two studies only described that there were no SAEs [30,36].
3.iv.5. Vitamin C Concentrations after Supplementation
In 8 of the studies, plasma or serum concentrations of vitamin C were measured [24,25,27,28,31,32,33,34]. In 4 of those studies, baseline values were mentioned past the authors [25,31,32,33]. In three of these studies, the plasma vitamin C concentrations earlier supplementation were normal to high, ranging betwixt 45 to 66 µM on average. In 14 of 22 patients with late-phase terminal cancer, the investigators describe low hateful baseline values, but write downwards that the mean plasma value was 100 µM earlier supplementation [33].
All viii studies mentioned plasma or serum vitamin C concentrations after the start of supplementation. Two studies dosed vitamin C in participants based on pinnacle plasma concentrations after IVC with a target range of twenty–23 mM. In 13 patients with ovarian cancer, it is not mentioned if this goal was always reached; in lx patients with various types of newly diagnosed cancer only 54% of patients reached the required level at the end of the handling period. Iii other articles as well depict acme plasma levels straight after IVC between 14 to xix.3 mM [25,28,31].
In 3 studies it is not explained at what moment in time plasma vitamin C concentrations were measured after supplementation. In the study in which mostly vitamin C scarce patients were treated, mean plasma levels during IVC were ane.one mM (range 0.38–3.0) [33]. In terminal cancer patients, the majority of the 1532 control patients had plasma concentrations of less than 85 µM, while the majority of the 294 vitamin C supplemented patients had concentrations higher than 114 µM [24]. In newly diagnosed breast cancer patients, researchers used oral supplementation in patients with normal baseline plasma vitamin C concentrations and measured values between 111 to 124 µM after supplementation [32].
In two studies information technology is mentioned that leukocyte vitamin C levels were measured [24,32], but in just 1 written report the results are given. In that study, the investigators did not run across a significant divergence in leukocyte vitamin C concentrations in 27 patients with newly diagnosed chest cancer before and after the start of oral vitamin C supplementation.
four. Discussion
4.1. Summary of the Main Results
This systematic review presents the results and quality assessment of 19 studies to evaluate the upshot of vitamin C treatment in cancer patients. Information technology is difficult to depict whatever conclusions, since these studies take a large variety of outcome measures and the included studies also differ in study population (from newly diagnosed to advanced cancer), co-interventions (none or various adjuvant anti-cancer treatments), and vitamin C treatment (dissimilar doses, schedules and modes of delivery). The results of some studies do advise that vitamin C might accept a positive event on overall survival, clinical response, QOL and PS. However, this effect cannot exist generalized to all patient groups with cancer. The best indication of a positive effect of vitamin C is seen in the RCT where it was used intravenously in elderly acute myeloid leukemia patients who were as well treated with decitabine. This outcome is near likely due to directly regulation of ten-eleven-translocation (TET) activity by vitamin C in synergy with decitabine. TET enzymes are dioxygenases that are important for Deoxyribonucleic acid methylation and are oft less functional in patients with AML. One of the potential working mechanisms of decitabine already is the upregulation of the TET proteins; it is idea that vitamin C enhances this result [37]. In all other studies, specially those that included patients with various types of cancer, the results were less clear. Therefore, it is not proven and seems quite unlikely that the pro-oxidant capacity of vitamin C in loftier dosage creates a positive effect on overall survival, clinical response, QOL or PS in cancer patients in general.
More half of the studies that researched QOL or PS saw a beneficial effect. However, since these studies were not blinded, patients may have experienced the well-known 'placebo consequence'. In this respect, the observation of Creagan et al. is of import, since he demonstrates a positive result in nearly 60% of the patients on placebo. These factors might have favored a positive outcome of vitamin C treatment in all studies, especially those in which event was assessed through self-administered questionnaires that are highly subjective.
The manner of delivery seems to be an of import factor in the effectiveness of the vitamin C treatment. In the studies with positive effects intravenous delivery was used, while the absence of effect was by and large after oral administration. This suggests that the vitamin C levels that can be reached by oral supplementation might not be high plenty for a possible effect, or that vitamin C is not captivated from the gastrointestinal tract.
In the 14 studies in which investigators reported on side effects of vitamin C, these side furnishings were mostly mild or none at all and could take been related to the cancer itself or to the concomitant therapy patients received. There was no obvious divergence in side effects of oral vitamin C compared to intravenous supplementation.
If measured at baseline, simply i of the studied patient groups was vitamin C deficient. Mayhap supplementation would exist more than efficient and useful in patient groups that indeed are deficient. Unfortunately, also not many vitamin C plasma concentrations afterwards supplementation were documented, but with oral supplementation plasma concentrations were much lower than with IVC.
4.2. Quality of the Evidence
The quality of the evidence is poor due to the lack of extensive double-blinded RCTs. There are just 4 modest RCTs that have been undertaken on this bailiwick, and in only 2 of these RCTs intravenous supplementation of vitamin C was used, which seems to be the most optimal to increment vitamin C concentrations in the patients.
All manufactures showed one or more forms of bias in the study procedures. Since only 4 studies were randomized, there is a high risk for selection bias. Particularly the absence of blinding procedures has resulted in functioning and detection bias. Result assessors might accept overestimated the furnishings of vitamin C in patients, based on knowledge of their characteristics and clinical status.
In general, virtually studies examined in this review show some positive effects of vitamin C supplementation on cancer. However, this could too be due to a publication bias of studies with a good result and are at best indicative that it is unlikely that vitamin C supplementation in any form is harmful in patients with cancer.
4.3. Potential Biases in the Review Process
Article option and information extraction was peer reviewed by a second author. Assessment of the quality of included studies was performed independently by two authors, with discrepancies resolved by discussion and a third writer when necessary. The authors therefore believe the review process was unbiased. The only limitation is the quality of the included studies.
v. Conclusions
The results of this review practise not prove that there is a clinically relevant positive issue of vitamin C supplementation in most cancer patients on the overall survival, clinical status, QOL and PS. The quality of the studies published is depression and the interventions and patient groups are very diverse. The best indication of a positive upshot is seen in acute myeloid leukemia patients in combination with decitabine, and in vitro data also show a synergistic efficacy of both treatments. An effect in some other patient groups might still be possible and might take been overlooked. In improver, clear pharmacological data might exist needed to optimize treatment plans.
Handling with vitamin C is likely to be safe, with near no serious adverse events and minimal mild side effects, even with high doses of intravenous supplementation. There are besides no indications that cancer progresses faster nether vitamin C supplementation.
We see the results of this review as an indication that it is safe to examine vitamin C supplementation farther in a randomized controlled setting. Therefore, we are planning to investigate the effect of vitamin C supplementation on immune recovery in patients that receive intensive chemotherapy and/or stem cell transplantation.
Supplementary Materials
The following are bachelor online at https://www.mdpi.com/2072-6643/xi/5/977/s1, Supplementary File S1: Literature Search, Supplementary File S2: Used take chances of bias tools, Supplementary File S3: Risk of bias summary.
Funding
This research received no external funding.
Conflicts of Involvement
The authors declare no conflict of involvement.
References
- Cameron, E.; Pauling, L. Supplemental ascorbate in the supportive treatment of cancer: Prolongation of survival times in concluding human cancer. Proc. Natl. Acad. Sci. Us 1976, 73, 3685–3689. [Google Scholar] [CrossRef] [PubMed]
- Cameron, E.; Campbell, A. The orthomolecular treatment of cancer. Ii. Clinical trial of high-dose ascorbic acrid supplements in advanced human being cancer. Chem. Biol. Interact. 1974, 9, 285–315. [Google Scholar] [CrossRef]
- Creagan, East.T.; Moertel, C.K.; O'Fallon, J.R.; Schutt, A.J.; O'Connell, M.J.; Rubin, J.; Frytak, South. Failure of high-dose vitamin c (ascorbic acid) therapy to benefit patients with advanced cancer. A controlled trial. N. Engl. J. Med. 1979, 301, 687–690. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Sun, H.; Wang, Y.; Riordan, H.D.; Hewitt, Due south.M.; Katz, A.; Wesley, R.A.; Levine, Grand. Vitamin c pharmacokinetics: Implications for oral and intravenous utilise. Ann. Intern. Med. 2004, 140, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Espey, Yard.Thou.; Dominicus, A.Y.; Pooput, C.; Kirk, K.L.; Krishna, G.C.; Khosh, D.B.; Drisko, J.; Levine, One thousand. Pharmacologic doses of ascorbate deed as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proc. Natl. Acad. Sci. The states 2008, 105, 11105–11109. [Google Scholar] [CrossRef] [PubMed][Greenish Version]
- Immature, J.I.; Zuchner, Due south.; Wang, 1000. Regulation of the epigenome past vitamin c. Annu. Rev. Nutr. 2015, 35, 545–564. [Google Scholar] [CrossRef] [PubMed]
- Kassouf, West.; Highshaw, R.; Nelkin, 1000.M.; Dinney, C.P.; Kamat, A.M. Vitamins C and K3 sensitize human urothelial tumors to gemcitabine. J. Urol. 2006, 176, 1642–1647. [Google Scholar] [CrossRef]
- Bober, P.; Alexovic, Thou.; Talian, I.; Tomkova, Z.; Viscorova, Z.; Benckova, Thou.; Andrasina, I.; Ciccocioppo, R.; Petrovic, D.; Adamek, M.; et al. Proteomic analysis of the vitamin C upshot on the doxorubicin cytotoxicity in the MCF-7 chest cancer jail cell line. J. Cancer Res. Clin. Oncol. 2017, 143, 35–42. [Google Scholar] [CrossRef]
- Jung, Southward.A.; Lee, D.H.; Moon, J.H.; Hong, South.W.; Shin, J.S.; Hwang, I.Y.; Shin, Y.J.; Kim, J.H.; Gong, E.Y.; Kim, S.Thou.; et al. L-Ascorbic acid can abrogate SVCT-2-dependent cetuximab resistance mediated by mutant KRAS in man colon cancer cells. Free. Radic. Biol. Med. 2016, 95, 200–208. [Google Scholar] [CrossRef]
- Kalita, S.; Verma, A.K.; Prasad, S.B. Chlorambucil and ascorbic acrid-mediated anticancer activity and hematological toxicity in Dalton's ascites lymphoma-bearing mice. Indian J. Exp. Biol. 2014, 52, 112–124. [Google Scholar]
- Kurbacher, C.G.; Wagner, U.; Kolster, B.; Andreotti, P.Due east.; Krebs, D.; Bruckner, H.W. Ascorbic acid (vitamin C) improves the antineoplastic action of doxorubicin, cisplatin, and paclitaxel in human breast carcinoma cells in vitro. Cancer Lett. 1996, 103, 183–189. [Google Scholar] [CrossRef]
- Huijskens, Yard.J.; Walczak, K.; Koller, Due north.; Briede, J.J.; Senden-Gijsbers, B.50.; Schnijderberg, M.C.; Bos, G.Chiliad.; Germeraad, W.T. Technical accelerate: ascorbic acrid induces development of double-positive T cells from human hematopoietic stalk cells in the absence of stromal cells. J. leukoc. biol. 2014, 96, 1165–1175. [Google Scholar] [CrossRef]
- Huijskens, Chiliad.J.; Walczak, M.; Sarkar, South.; Atrafi, F.; Senden-Gijsbers, B.Fifty.; Tilanus, M.G.; Bos, 1000.M.; Wieten, L.; Germeraad, W.T. Ascorbic acid promotes proliferation of natural killer cell populations in civilization systems applicable for natural killer cell therapy. Cytotherapy 2015, 17, 613–620. [Google Scholar] [CrossRef]
- Van Gorkom, G.N.Y.; Klein Wolterink, R.Chiliad.J.; Van Elssen, C.; Wieten, L.; Germeraad, W.T.V.; Bos, G.M.J. Influence of Vitamin C on Lymphocytes: An Overview. Antioxidants 2018, 7, 41. [Google Scholar] [CrossRef]
- Huijskens, K.J.; Wodzig, W.K.; Walczak, M.; Germeraad, W.T.; Bos, G.Thou. Ascorbic acid serum levels are reduced in patients with hematological malignancies. Result. Immun. 2016, half dozen, eight–10. [Google Scholar] [CrossRef][Green Version]
- Mayland, C.R.; Bennett, One thousand.I.; Allan, K. Vitamin C deficiency in cancer patients. Palliat. Med. 2005, nineteen, 17–20. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.Grand.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009, vi, e1000100. [Google Scholar] [CrossRef]
- Cochrane Consumers and Communication. Information Extraction Template for Included Studies. Version 1.viii, updated twenty November 2016. Bachelor online: http://cccrg.cochrane.org/author-resources (accessed on 1 August 2018).
- Higgins, J.P.; Altman, D.Chiliad.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, Thou.F.; Weeks, Fifty.; Sterne, J.A. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Higgins, J.P.T.; Reeves, B.C.; On behalf of the development group for ACROBAT-NRSI. A Cochrane Risk of Bias Cess Tool: For Non-Randomized Studies of Interventions (ACROBAT-NRSI), Version one.0.0 24 September 2014. Available online: http://world wide web.bristol.ac.uk/media-library/sites/social-community-medicine/images/centres/cresyda/ACROBAT-NRSI%20Version%201_0_0.pdf (accessed on one August 2018).
- West, S.; King, 5.; Carey, T.S.; Lohr, K.Northward.; McKoy, Northward.; Sutton, S.F.; Lux, 50. Systems to rate the force of scientific evidence. Evid. Rep. Technol. Assess. 2002, 47, 1–11. [Google Scholar]
- Bazzan, A.J.; Zabrecky, G.; Wintering, Due north.; Newberg, A.B.; Monti, D.A. Retrospective Evaluation of Clinical Experience With Intravenous Ascorbic Acid in Patients With Cancer. Integr. Cancer Ther. 2018, 17, 912–920. [Google Scholar] [CrossRef]
- Cameron, East.; Pauling, 50. Supplemental ascorbate in the supportive handling of cancer: Reevaluation of prolongation of survival times in terminal man cancer. Proc. Natl. Acad. Sci. Us 1978, 75, 4538–4542. [Google Scholar] [CrossRef][Greenish Version]
- Cameron, E.; Campbell, A. Innovation vs. quality control: an 'unpublishable' clinical trial of supplemental ascorbate in incurable cancer. Med. Hypotheses 1991, 36, 185–189. [Google Scholar] [CrossRef]
- Hoffer, L.J.; Robitaille, L.; Zakarian, R.; Melnychuk, D.; Kavan, P.; Agulnik, J.; Cohen, V.; Small-scale, D.; Miller, W.H. High-dose intravenous vitamin C combined with cytotoxic chemotherapy in patients with advanced cancer: a stage I-Two clinical trial. PloS ONE 2015, 10, e0120228. [Google Scholar] [CrossRef]
- Gunes-Bayir, A.; Kiziltan, H.S. Palliative Vitamin C Application in Patients with Radiotherapy-Resistant Os Metastases: A Retrospective Written report. Nutr. Cancer 2015, 67, 921–925. [Google Scholar] [CrossRef]
- Ma, Y.; Chapman, J.; Levine, Grand.; Polireddy, Thou.; Drisko, J.; Chen, Q. Loftier-dose parenteral ascorbate enhanced chemosensitivity of ovarian cancer and reduced toxicity of chemotherapy. Sci. Transl. Med. 2014, vi, 222ra218. [Google Scholar] [CrossRef]
- Mikirova, Northward.; Casciari, J.; Rogers, A.; Taylor, P. Effect of high-dose intravenous vitamin C on inflammation in cancer patients. J. Transl. Med. 2012, x, 189. [Google Scholar] [CrossRef]
- Moertel, C.Yard.; Fleming, T.R.; Creagan, E.T.; Rubin, J.; O'Connell, M.J.; Ames, K.M. Loftier-dose vitamin C versus placebo in the treatment of patients with advanced cancer who have had no prior chemotherapy. A randomized double-blind comparison. The New England journal of medicine. N. Engl. J. Med. 1985, 312, 137–141. [Google Scholar] [CrossRef]
- Murata, A.; Morishige, F.; Yamaguchi, H. Prolongation of survival times of terminal cancer patients past administration of large doses of ascorbate. Int. J. Vitam. Nutr. Res. 1982, 23, 103–113. [Google Scholar]
- Nielsen, T.G.; Hojgaard, M.; Andersen, J.T.; Jorgensen, N.R.; Zerahn, B.; Kristensen, B.; Henriksen, T.; Lykkesfeldt, J.; Mikines, K.J.; Poulsen, H.E. Weekly ascorbic acrid infusion in castration-resistant prostate cancer patients: A single-arm stage Ii trial. Transl. Androl. Urol. 2017, half dozen, 517–528. [Google Scholar] [CrossRef]
- Poulter, J.Chiliad.; White, W.F.; Dickerson, J.W. Ascorbic acrid supplementation and five year survival rates in women with early chest cancer. Acta. Vitaminol. Enzymol. 1984, 6, 175–182. [Google Scholar]
- Riordan, H.D.; Casciari, J.J.; Gonzalez, M.J.; Riordan, Northward.H.; Miranda-Massari, J.R.; Taylor, P.; Jackson, J.A. A airplane pilot clinical written report of continuous intravenous ascorbate in concluding cancer patients. Puerto Rico Wellness Sci. J. 2005, 24, 269–276. [Google Scholar]
- Takahashi, H.M.H.; Ynagisawa, A. Loftier-dose intravenous vitamin C improves quality of life in cancer patients. Pers. Med. Univ. 2012, 1, 49–53. [Google Scholar] [CrossRef]
- Vollbracht, C.; Schneider, B.; Leendert, Five.; Weiss, G.; Auerbach, L.; Beuth, J. Intravenous vitamin C administration improves quality of life in breast cancer patients during chemo-/radiotherapy and aftercare: results of a retrospective, multicentre, epidemiological cohort written report in Germany. In Vivo 2011, 25, 983–990. [Google Scholar]
- Yeom, C.H.; Jung, G.C.; Song, K.J. Changes of final cancer patients' health-related quality of life later on high dose vitamin C assistants. J. Korean Med. Sci. 2007, 22, 7–xi. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhu, H.; Huang, J.; Zhu, Y.; Hong, Yard.; Zhu, H.; Zhang, J.; Li, S.; Yang, L.; Lian, Y.; et al. The synergy of Vitamin C with decitabine activates TET2 in leukemic cells and significantly improves overall survival in elderly patients with astute myeloid leukemia. Leuk. Res. 2018, 66, i–7. [Google Scholar] [CrossRef] [PubMed]
Effigy 1. Flow diagram of the article choice.
Figure ane. Catamenia diagram of the commodity choice.
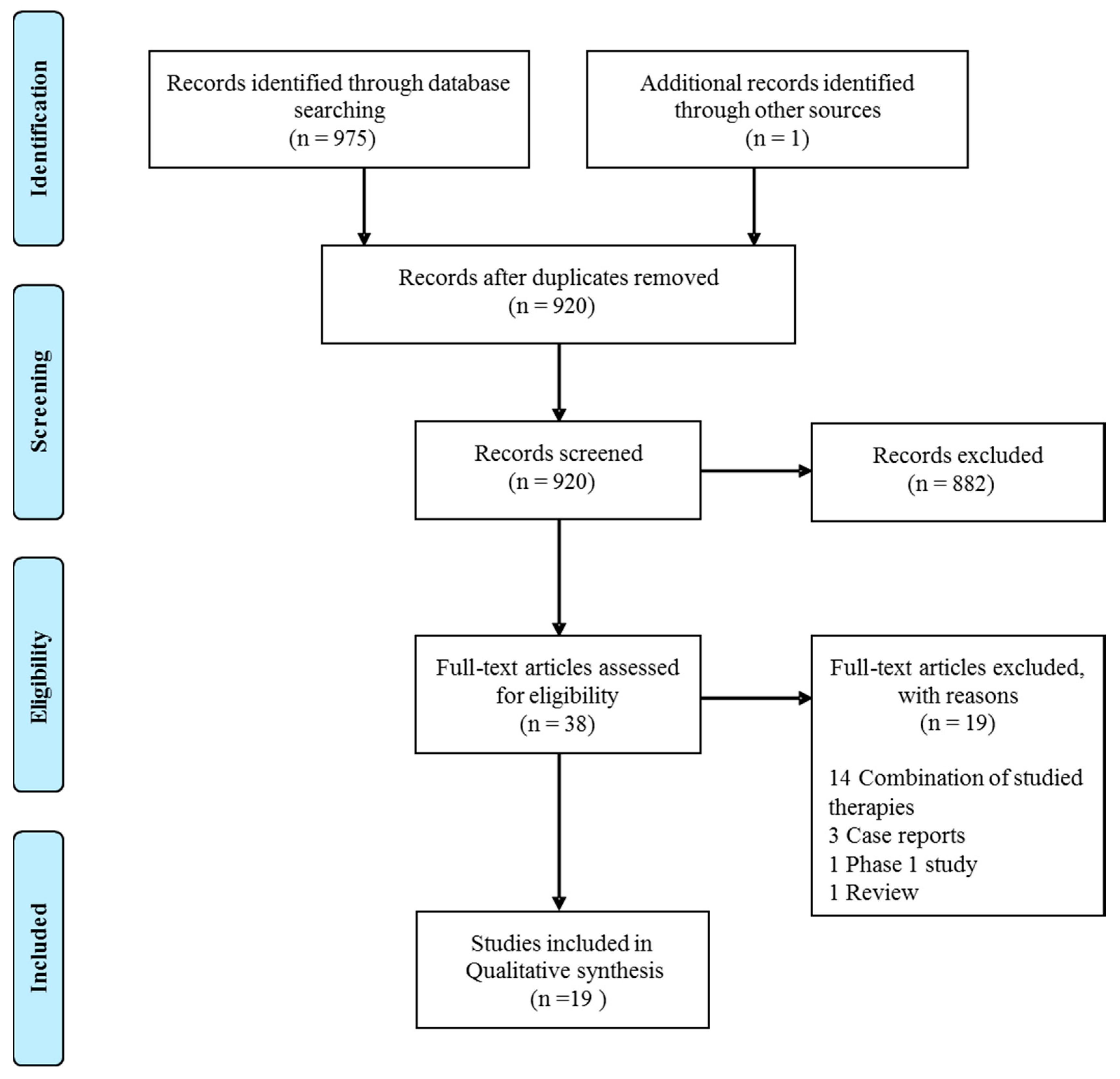
Figure two. Itemized Judgments for risk of bias based on "The Cochrane Collaboration's tool for assessing take a chance of bias in randomized trials". Adventure is presented percentages beyond all included RCTs.
Figure two. Itemized Judgments for gamble of bias based on "The Cochrane Collaboration's tool for assessing gamble of bias in randomized trials". Gamble is presented percentages across all included RCTs.
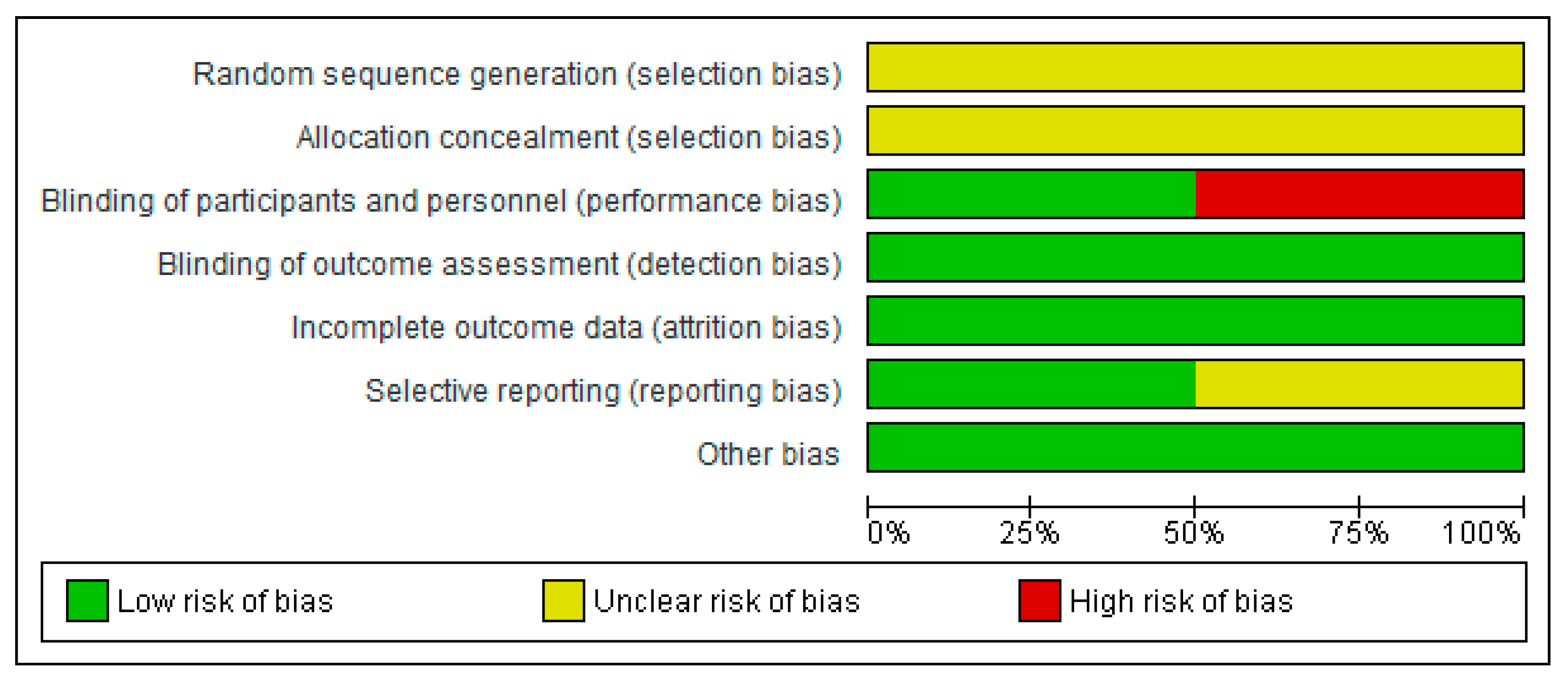
Table 1. Characteristics and outcome of the included studies in alphabetical order.
Table 1. Characteristics and outcome of the included studies in alphabetical order.
| Written report | N(Vitamin C/ controls) | Type | Participants | Interven-tion | Concomitant therapy | Principal endpoints | Adverse events |
|---|---|---|---|---|---|---|---|
| Bazzan 2018 [22] | 86 (86/0) | Retrospective cohort | All types of cancer in dissimilar settings | 50 - 150 g 4, at least 5 times (total 3034 doses) | 32 patients none, 54 patients chemotherapy | 20 of 40 patients comeback of fatigue, 15 of 86 patients improvement of appetite. | Mostly balmy adverse events (AEs), like nausea, airsickness and discomfort at injection side (<3% of infusions). Self- limiting to time of infusions. No serious AEs (SAEs) related to vitamin C |
| Cameron 1974 [2] | 50(50/0) | Retrospective accomplice | Advanced stage cancer patients | ten g a twenty-four hours IV for 10 days + oral vitamin C | None | 10 minimal response, eleven growth retardation, three stable affliction, five tumor regression. Less pain, reduction in ascites/pleural effusions. | Fluid retention, edema, bitchy symptoms, tumor hemorrhage/necrosis. |
| Cameron 1976 [1] | 1100(100/1000) | Case-control | Incurable cancer patients | 10 g a day 4 for x days + oral vitamin C | Conventional anti-cancer treatment | Mean overall survival (OS) 210 days vs. 50 days in controls (4.2x more) | Non reported (NR) |
| Cameron 1978 [23] | 1100(100/grand) | Case-control | Concluding cancer patients | 10 grand a day IV for x days + oral vitamin C | None | Recalculation of Cameron 1976. Average Os (seven.7x more = 288 days) | NR |
| Cameron 1991 [24] | 1826(294/1532) | Instance-command | Concluding cancer patients | 10 one thousand a mean solar day Iv for x days + oral vitamin C | None | Bone 343 days vs. 180 days in controls | NR |
| Creagan 1979 [3] | 123(sixty/63) | RCT | Advanced stage cancer patients | 10 k a day orally | NR | Identical survival.Operation status (PS) identical. 58% vs. 63% some improvement in symptoms | Nausea, vomiting |
| Hoffer 2015 [25] | 14 (14/0) | Uncontrolled phase 2 | Advanced stage cancer patients | ane.5g/kg IV 2-3 times per calendar week. | Chemothera-py | vi transient, partly long-lasting stable affliction | Edema, thirst, nausea, vomiting, headache, chills |
| Günes-Bayir 2015 [26] | 39 (15/24) | Example-control | Bone metastases from various types of cancer | 2.v chiliad IV a solar day | NR | OS 10 months vs. 2 months in controls. Decrease in hurting in 9/15 vs. 5/24 in controls. PS comeback in 4/fifteen vs. 1/24 in controls. | 40% balmy diarrhea, 30% mild oliguria |
| Ma 2014 [27] | 25 (13/12) | RCT | Newly diagnosed stage III and IV ovarian cancer after debulking | IV ii times per week using a dose escalating protocol (concluding dose either 75 or 100g) for 12 months. | Paclitaxel and carboplatin chemotherapy | Trend in improvement OS, 25.5 months vs. 16.75 months in controls, (not significant). | Fewer chemotherapy related side effects with vitamin C, no relevant AEs of vitamin C. |
| Mikirova 2012 [28] | 45 (45/0) | Retrospective cohort | Various types of cancer, mostly metastatic | Escalate to fifty g 4 3 times per calendar week for a median of ix times | NR | 76% reduction in C –reactive protein, 75% reduction in prostate-specific antigen (PSA) | NR |
| Moertel 1985 [29] | 100 (51/49) | RCT | Avant-garde colorectal cancer | 10 g a 24-hour interval orally | None | Median Bone 2.9 months vs. 4.i months in controls. 7/11 symptom relief vs. 11/17 in controls | Low incidence of AEs, mild. |
| Murata 1982 [30] | 130 (111/nineteen) | Not randomized clinical trial | Final cancer patients | Site 1: 6–30 g a day orally and 10–20g IV. Site 2: 0.five–3 1000 or 5–30 g per twenty-four hour period orally. | NR | Site 1: average OS with high dose vitamin C 246 days vs. 43 days with low dose.Site 2: average OS with high dose vitamin C 115 days vs. 48 days in controls. Less utilise of narcotic drugs in vitamin C treated patients: 17% in loftier dose vs. l% in low dose vs. 79% in controls. Improved state of wellbeing, improved appetite, increased mental alacrity | No SAEs |
| Nielsen 2017 [31] | 23 (23/0) | Uncontrolled phase II | Chemothe-rapy-naïve metastatic castration-resistant prostate cancer | Weekly infusions for 12 weeks. Calendar week 1: 5 g, week2: 30 thou, week 3–12: 60 chiliad + oral 500 mg/day. | None | 75% of patients PSA increase at 12 weeks, ane PSA decrease of 27%. 80% unchanged PS at calendar week 12, ii improved, 2 worse score.Quality of life (QOL) identical baseline to week 12. | 53 AE, mostly balmy and not related to vitamin C. 11 SAEs, explained by progression of prostate cancer. 2 pulmonary embolisms. |
| Poulter 1984 [32] | 66 (27/25) | Non randomized clinical trial | Newly diagnosed breast cancer | 3 grand a day orally | NR | No alter in survival rates | NR |
| Riordan 2005 [33] | 24 (24/0) | Uncontrolled phase Ii | Late stage terminal cancer, mostly colorectal (19) | 150 to 710 mg/kg/24-hour interval IV for 8 weeks | None | 1 patient stable disease, all others progressive disease. | Mild: nausea (46%), edema (29%), dry mouth or skin (29%), fatigue (25%). Serious: 1 kidney stones, 1 hypokalemia. |
| Takaha-shi 2012 [34] | 60 (60/0) | Prospective cohort | Newly diagnosed cancer of various types | Four dose of 12.5–15 g, 25 m and 50 g + vitamin C orally 2–iv 1000 a 24-hour interval. | Chemothera-py (due north = 33), radiation therapy (n = 1), none (n = 2) | Comeback in QOL: score 44.6 before treatment vs. 53.ii at 2 weeks and 61.4 at 4 weeks. | Balmy (grade 1), most often headache (8.3%), nausea (eight.three%) |
| Voll-bracht 2011 [35] | 125 (53/72) | Retrospective accomplice | Chest cancer stages IIa–IIIb. | IV vii.5 g one time a week for at least 4 weeks | Primary surgical treatment +/- adjuvant chemotherapy +/- adjuvant radiotherapy | Reduced QOL related side furnishings, slight increment PS during adjuvant treatment (80% vs. 71%) and aftercare (87% vs. 78%). | None |
| Yeom 2007 [36] | 39 (39/0) | Uncontrolled stage 2 | Terminal cancer of diverse types, stage IV. | 10 g IV twice + 4 g oral vitamin C daily for a week. | NR | Wellness score improved from 3637 to 5537 after vitamin C. | No vitamin C supplementation stopped considering of side effects. |
| Zhao 2017 [37] | 73 (39/34) | RCT | Newly diagnosis elderly with acute myeloid leukemia | 50–lxxx mg/kg Four during 10 days/month, at most 10 months | Decitabine, cytarabine and aclarubicin chemotherapy | Median OS 15.3 months vs. 9.three months in controls (p = 0.039). Complete remission rate higher with Vitamin C: 84.6% vs. 70.6% after 2 courses. | Identical corporeality of AEs and SAEs in both groups. |
Table 2. Take chances-of –bias assessment of comparative studies. Judgment for take a chance of bias based on the ROBINS-I tool for each included comparative study (high, moderate, low).
Table 2. Risk-of –bias assessment of comparative studies. Judgment for risk of bias based on the ROBINS-I tool for each included comparative study (loftier, moderate, low).
| Report | Evidence of Selection Bias/Prognostic Imbalance | Bias Due to Misreckoning Factors | Bias in Measurement of Outcomes | Follow-up of Participants Sufficiently Consummate | Bias Due to Choice of Reported Results or due to Missing Information | Comparability of Cohorts on Important Confounding Factors |
|---|---|---|---|---|---|---|
| Cameron 1976 [1] | Loftier take chances | High hazard | Moderate risk | Moderate take a chance | Depression risk | Moderate adventure |
| Cameron 1978 [23] | High chance | High chance | Moderate risk | Moderate adventure | Low take a chance | Moderate run a risk |
| Cameron 1991 [24] | Moderate gamble | High risk | Low risk | Moderate risk | Depression risk | Moderate adventure |
| Günes-Bayir 2015 [26] | Moderate hazard | Moderate adventure | Moderate hazard | Moderate risk | Low hazard | Moderate risk |
| Murata 1982 [xxx] | High risk | High risk | Moderate risk | Moderate risk | Moderate take chances | Moderate risk |
| Poulter 1984 [32] | High run a risk | Moderate risk | Moderate take a chance | High take chances | Moderate take a chance | Moderate take chances |
| Vollbracht 2011 [35] | High risk | Moderate risk | Moderate gamble | Depression risk | Depression adventure | Loftier gamble |
Table 3. Quality assessment of non-comparative studies. Judgment for quality of the studies based on the Effective Public Health Practice Project tool for each included non-comparative study (stiff, moderate, weak).
Tabular array 3. Quality cess of non-comparative studies. Judgment for quality of the studies based on the Constructive Public Wellness Practice Projection tool for each included non-comparative written report (stiff, moderate, weak).
| Study | Pick | Study Design | Confounders | Blinding | Information Drove Methods | Withdrawals and Drop-Outs |
|---|---|---|---|---|---|---|
| Bazzan 2018 [22] | Weak | Weak | Weak | Weak | Weak | Not applicable |
| Cameron 1974 [ii] | Moderate | Weak | Weak | Weak | Moderate | Not applicable |
| Hoffer 2015 [25] | Moderate | Weak | Weak | Weak | Strong | Strong |
| Mikirova 2012 [28] | Weak | Weak | Weak | Weak | Moderate | Strong |
| Nielsen 2017 [31] | Moderate | Weak | Moderate | Weak | Strong | Strong |
| Riordan 2005 [33] | Moderate | Weak | Weak | Weak | Strong | Stiff |
| Takahashi 2012 [34] | Moderate | Weak | Weak | Weak | Moderate | Strong |
| Yeom 2007 [36] | Moderate | Weak | Weak | Weak | Moderate | Strong |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open admission commodity distributed under the terms and conditions of the Creative Commons Attribution (CC Past) license (http://creativecommons.org/licenses/past/4.0/).
Source: https://www.mdpi.com/2072-6643/11/5/977/htm
0 Response to "Intravenous Vitamin C and Cancer a Systematic Review"
Post a Comment